Subperiosteal Implants
- Home
- Treatments
- Subperiosteal Implants
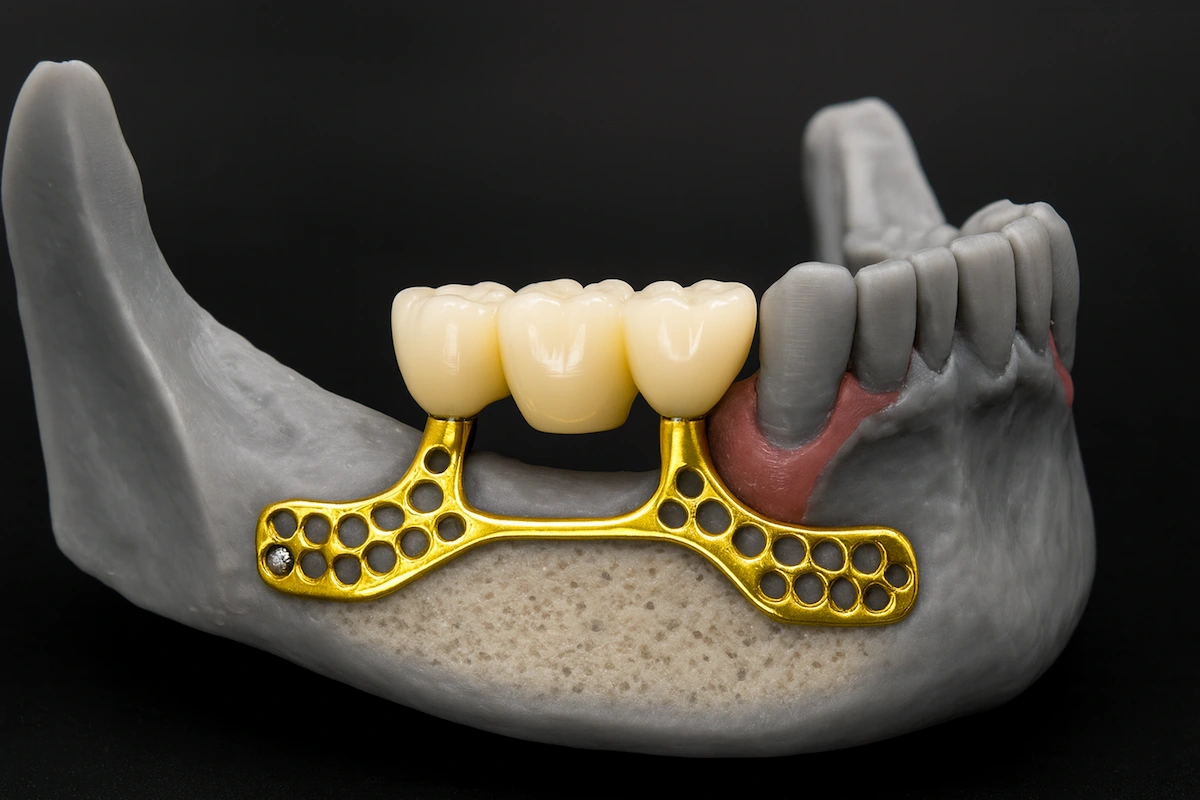
A subperiosteal implant is a custom-designed metal framework that sits on top of the jawbone, beneath the gum tissue, rather than being inserted into the bone like a conventional implant. It is an established alternative for patients with advanced bone loss who are not suitable candidates for standard endosseous implants. At Vitrin Clinic, subperiosteal implants are planned using precise 3D imaging to ensure an accurate fit and a stable, long-term result.
Treatment Process
A clear step-by-step overview of how the treatment is planned and performed, from the initial consultation to the final results, ensuring comfort, safety, and predictable outcomes.
01
Initial Assessment & Imaging
We begin with a full clinical examination and CBCT scan to evaluate your bone volume, density, and anatomy. This imaging is essential to determine whether a subperiosteal implant is the right solution for your case.
02
Custom Framework Design
Using your 3D scan data, a custom titanium framework is digitally designed to conform precisely to the shape of your jawbone. This bespoke fit is what gives subperiosteal implants their stability without requiring bone integration.
03
Surgical Placement
The framework is placed in a minor surgical procedure under general anaesthesia. The implant is positioned on top of the jawbone and secured beneath the gum tissue, with posts protruding through the gum to support the final restoration.
04
Healing & Integration
The gum tissue heals around the framework over a period of several weeks. During this phase, temporary restorations may be provided to maintain function and aesthetics while healing progresses.
05
Final Restoration
Once healing is confirmed, your permanent prosthetic teeth, whether a crown, bridge, or full arch restoration, are attached to the implant posts. Final adjustments are made to ensure comfort, bite accuracy, and a natural appearance.
What Is a Subperiosteal Implant?
A subperiosteal implant (SPI) is a custom-fabricated titanium framework positioned on the surface of the alveolar bone, beneath the periosteum the fibrous membrane covering the bone rather than within it. Unlike conventional endosseous implants, which require sufficient bone volume for osseointegration via direct bone-to-implant contact, a subperiosteal implant rests on the existing cortical bone surface and achieves its retention through periosteal healing and the anatomical contouring of the framework to the patient's bone topography.
The subperiosteal concept is not new it was first described in the 1940s but the modality has undergone a fundamental clinical renaissance through digital technology. Contemporary subperiosteal implants are designed entirely from the patient's cone beam computed tomography (CBCT) data using computer-aided design (CAD) software, and manufactured from medical-grade titanium alloy (Ti-6Al-4V) using either additive manufacturing (direct metal laser sintering DMLS, or selective laser melting SLM) or subtractive CAD/CAM milling. This digital workflow eliminates the imprecision of historical impression-based techniques and produces a framework that fits the patient's bone with micron-level accuracy before surgery.
97.8% Implant Survival Rate | 3 Clinic Visits Required | No Bone Grafting Required | CBCT + CAD Digital Design Workflow |
|---|---|---|---|
Clinical Indications: Who Is a Candidate?
The subperiosteal implant is indicated for a specific and well-defined patient population: those with severe alveolar bone atrophy for whom conventional endosseous implants are not feasible without extensive and high-risk bone augmentation procedures. Accurate patient selection based on thorough radiographic and clinical assessment is the primary determinant of treatment success.
Severe Alveolar Ridge Atrophy
Severe horizontal and vertical resorption of the alveolar ridge classified as Cawood and Howell Class V or VI leaves insufficient bone volume for standard-diameter endosseous implants. Residual bone height below 5 to 6 mm in the posterior mandible or maxilla, knife-edge ridges, and extensive pneumatisation of the maxillary sinus in the posterior maxilla are the primary anatomical presentations. A subperiosteal implant resolves this by using the existing cortical bone surface as its foundation rather than requiring bone volume that is not present.
Refusal or Contraindication to Bone Grafting
Patients who decline extensive bone augmentation procedures including block grafts, sinus lifts, or guided bone regeneration with long healing periods are candidates for subperiosteal implant treatment. Patients for whom bone augmentation carries elevated systemic risk due to anticoagulant therapy, bisphosphonate use, osteoporosis, or compromised healing capacity may also be better managed with a subperiosteal approach that avoids the need for large-volume grafting and its associated morbidity.
Proximity to Critical Anatomical Structures
In cases where the inferior alveolar nerve canal, the mental foramen, or the floor of the maxillary sinus precludes safe endosseous implant placement at adequate dimensions even with minor augmentation a subperiosteal framework can be designed around these structures rather than requiring repositioning surgery. The three-dimensional CAD design allows the engineer and surgeon to map the framework precisely relative to the CBCT-identified neurovascular anatomy before fabrication.
Complete or Extensive Edentulism with Atrophy
A full-arch subperiosteal framework supporting a fixed prosthesis across the entire dental arch is the most common treatment configuration. It replaces the conventional All-on-4 or All-on-6 approach for patients in whom the bone volume required for those techniques is not available. A single subperiosteal framework can support a complete fixed dental prosthesis from as few as four abutment emergence points, eliminating the need for zygomatic implants or complex sinus augmentation in many presentations.
Posterior Segment Atrophy Partial Rehabilitation
In partially edentulous patients with posterior segment atrophy particularly in the posterior mandible where residual bone height above the inferior alveolar nerve is severely reduced a partial subperiosteal framework can replace two to four missing posterior teeth without the sinus elevation or nerve repositioning that endosseous implant placement would require. A peer-reviewed case series from Policlinico Gemelli in Rome confirmed predictable clinical outcomes for custom subperiosteal frameworks in the posterior atrophic mandible of elderly patients over one year of follow-up.
When a Subperiosteal Implant Is Not Indicated
Subperiosteal implants are not appropriate for patients with active or uncontrolled periodontal disease, uncontrolled diabetes (HbA1c above 8%), or active oral infection at the planned surgical site. Active smoking significantly increases the risk of soft tissue dehiscence and partial framework exposure the most frequent complication reported in published series, occurring in 25.6% of cases in the 2024 systematic review. Where conventional endosseous implants are clinically feasible, they remain the evidence-based first choice. Subperiosteal treatment is a precision solution for a defined clinical problem, not a universal alternative to standard implantology.
Framework Design and Materials
The clinical performance of a subperiosteal implant is determined by the precision of its fit to the underlying cortical bone and the biological compatibility of its material. Both are products of the digital design workflow and both must be addressed explicitly in the surgical and prosthodontic planning process before fabrication.
Titanium Cortical Framework
The body of the subperiosteal implant is a three-dimensional titanium lattice or solid framework designed to conform precisely to the contours of the patient's residual cortical bone as captured in the CBCT scan. The framework rests on the bone surface and is secured with titanium mini-screws or fixation pins at defined cortical anchorage points to prevent micro-movement during the healing phase. The periosteum heals over the framework, incorporating it into the soft tissue envelope and achieving biological stabilisation through fibrous attachment rather than osseointegration.
Medical-grade titanium Grade 4 (commercially pure) or Ti-6Al-4V alloy is used for subperiosteal frameworks the same materials validated across decades of endosseous implant research for biocompatibility, corrosion resistance, and mechanical fatigue resistance. A randomised clinical trial from Cairo University comparing milled versus 3D-printed patient-specific subperiosteal frameworks in atrophic mandibular ridges found both fabrication methods produced comparable implant survival and placement accuracy at follow-up, with angular deviations of less than 2° between planned and actual framework position.
Abutments and Emergence Profile
The prosthetic abutments are integral components of the subperiosteal framework, designed simultaneously with the cortical body in the CAD software. Their number, position, angulation, and height are determined by the prosthetic plan establishing where the prosthesis will emerge through the gingival tissue and how occlusal forces will be distributed across the framework at function.
The abutment positions are designed to provide a passive fit for the prosthesis, appropriate emergence profile angles for hygiene access, and a biomechanically balanced load distribution across the framework and the underlying cortical bone. A diagnostic wax-up or digital prosthetic preview is produced before the framework is fabricated, so the prosthetic outcome is confirmed in the CAD environment before the implant is manufactured.
Cortical Anchorage Screws
Following surgical placement of the framework on the cortical bone surface, titanium mini-screws are inserted through pre-planned fixation points in the framework into the underlying cortical bone. These screws immobilise the framework during the initial periosteal healing phase preventing micro-movement that would disrupt fibrous integration. The number and position of fixation screws are determined in the CAD plan based on the available cortical bone thickness and the anticipated load distribution of the framework at function.
In some protocols, the fixation screws are subsequently removed at the second surgical visit once periosteal healing is confirmed. In others, they are designed to remain permanently as additional retention elements. The protocol is selected based on the framework design and the individual patient's healing trajectory.
CBCT → CAD → Fabrication → Surgery
The modern subperiosteal workflow is fully digital from imaging to implant. CBCT DICOM data is imported into CAD software (Exocad, 3Shape, or equivalent), the cortical bone surface is segmented, the framework is designed over the segmented bone surface, abutment positions are confirmed against the prosthetic wax-up, and the confirmed design is exported as an STL file for manufacturing. This digital chain eliminates the dimensional inaccuracies inherent in the original impression-based subperiosteal technique and is the primary reason for the improved survival rates reported in contemporary literature compared to historical series.
Subperiosteal Implant vs. Alternative Approaches for Atrophic Jaws
For patients with severe alveolar atrophy, multiple treatment pathways exist each with distinct advantages, risks, treatment timelines, and biological requirements.
The following comparison is compiled from peer-reviewed systematic reviews and clinical outcome studies.
Criterion | Subperiosteal Implant | Bone Graft + Endosseous Implant | Zygomatic Implants | Implant-Supported Overdenture |
|---|---|---|---|---|
Bone Volume Required | None rests on existing cortical surface | Substantial augmentation required first | None in alveolus zygomatic bone used | Minimal 2–4 short or narrow implants |
Bone Grafting Required | No eliminates graft phase entirely | Yes block, particulate, or sinus lift | No zygomatic anchorage bypasses alveolus | Sometimes mini-implants may be feasible |
Total Treatment Timeline | 3–6 months 3 clinic visits | 12–24 months graft healing + implant osseointegration | 4–6 months no graft, but complex surgery | 4–8 months implant osseointegration required |
Surgical Complexity | Moderate mucoperiosteal flap + framework placement | High augmentation + staged implant surgery | Highest zygomatic bone engagement, specialist-only | Lower minimal implants, often flapless |
Fixed Prosthesis Possible | Yes full fixed arch supported by framework | Yes once osseointegration confirmed | Yes fixed immediate or delayed loading | No overdenture is removable |
Primary Mechanism of Retention | Periosteal healing + cortical anchorage screws not osseointegration | Full osseointegration direct bone-to-implant contact | Osseointegration in zygomatic cortical bone | Osseointegration of supporting implants |
Main Clinical Risk | Partial framework exposure (25.6% in systematic review) manageable | Graft failure, infection, implant loss in grafted site | Sinusitis, oroantral communication, specialist dependency | Peri-implant disease, overdenture maintenance |
Patient Morbidity | Lower than graft + implant single surgical phase | High two or more surgical phases, donor site morbidity | Moderate single surgery but high-complexity anatomy | Low minor surgical intervention |
Compiled from: University of the Basque Country (UPV/EHU) — Additively Manufactured Subperiosteal Implants Systematic Review, PMC10844163, 2024 · NIH/PMC — Clinical Performance of Subperiosteal Implants in Full-Arch Rehabilitation: Systematic Review and Metanalysis, PMC12191889, 2025 · PMC11122366 — Long-Term Clinical Outcomes of 3D-Printed Subperiosteal Titanium Implants: A 6-Year Follow-Up.
Why Clinical Approach Matters
The clinical success of a subperiosteal implant is almost entirely determined by the precision of the pre-surgical digital workflow. Unlike endosseous implants where implant position can be adjusted within the bone at surgery a subperiosteal framework is fabricated to a fixed geometry before the patient enters the operating theatre. If the CBCT scan is of insufficient resolution, the digital segmentation of the bone surface is inaccurate, or the CAD design does not correctly account for the planned prosthetic outcome, the framework will not seat passively and a non-passive fit is the primary intraoperative complication requiring framework modification or, in severe cases, a new fabrication cycle.
The most consistently reported post-surgical complication in subperiosteal implant literature is partial framework exposure through the overlying soft tissue occurring in 25.6% of implants in the 2024 University of the Basque Country systematic review. This complication is strongly associated with inadequate soft tissue closure at surgery, thin gingival biotype, smoking, and insufficient space between the framework and the mucogingival junction. It is largely preventable with meticulous flap design, passive closure, and pre-surgical evaluation of the soft tissue envelope. At Vitrin Clinic, soft tissue assessment is a mandatory component of the pre-surgical planning protocol not an intraoperative consideration.
The digital design workflow has fundamentally transformed subperiosteal implantology. A 2024 randomised clinical trial from Cairo University comparing CAD/CAM milled and 3D-printed subperiosteal frameworks found angular deviations of less than 2° between the planned and achieved framework position in both groups, confirming that modern digital fabrication achieves clinically acceptable positional accuracy regardless of manufacturing method. This precision is the principal reason contemporary survival rates (97.8%) are substantially higher than those reported for impression-based historical techniques.
What the research shows
97.8% | 25.6% | <2° |
|---|---|---|
Implant survival rate of additively manufactured CAD/CAM subperiosteal implants at a weighted mean follow-up of 21.4 months across 227 implants in 227 patients confirming the clinical viability of the modern digital subperiosteal approach for severely atrophic jaw rehabilitation. | Rate of partial framework exposure through the overlying soft tissue the most frequent complication reported in the 2024 systematic review, occurring in 58 of 227 implants. Partial exposure is manageable in most cases but underscores the clinical importance of meticulous flap design, passive closure, and pre-surgical soft tissue assessment. | Angular deviation between planned and achieved subperiosteal framework position in a randomised clinical trial comparing CAD/CAM milled and 3D-printed titanium frameworks confirming that modern digital fabrication achieves clinically acceptable positional accuracy irrespective of manufacturing method. |
University of the Basque Country (UPV/EHU) Additively Manufactured SPI Systematic Review, PMC10844163, 2024 · PROSPERO CRD42023424211 | University of the Basque Country (UPV/EHU) PMC10844163 · Anitua E, Eguia A et al., Int J Implant Dent, 2024 | Cairo University Milled vs. 3D-Printed Patient-Specific Subperiosteal Implants RCT, PMC11979865, 2025 |
Vitrin Clinic Standard
Three clinic visits defined clinical objective at each
Visit One: CBCT, scanning, and framework design confirmation. Visit Two: surgical placement and provisional restoration. Visit Three: definitive prosthetic restoration. Each visit has a fixed clinical objective no visit is a filler appointment.
Prosthetic outcome confirmed before fabrication
The framework is not fabricated until the patient has reviewed and approved the digital design and the planned prosthetic outcome in the CAD environment. No titanium is printed until the design is clinically confirmed.
Passive fit verified before fixation
The framework is seated and passive fit confirmed at all contact points before fixation screws are placed. A framework that does not seat passively is not secured it is returned for adjustment or refabrication as clinically required.
Soft tissue complication risk assessed and communicated before surgery
Partial framework exposure risk the most common complication in published literature is assessed pre-surgically based on your tissue biotype, keratinized tissue width, and smoking status. You are informed of your specific risk profile before any commitment to surgical treatment.
Clinical Evidence & References
University of the Basque Country (UPV/EHU) & UIRMI, Spain
Clinical Performance of Additively Manufactured Subperiosteal Implants: A Systematic Review
International Journal of Implant Dentistry · February 2024Anitua E, Eguia A, Staudigl C, Alkhraisat MH · DOI: 10.1186/s40729-024-00521-6 · PMC10844163 · PROSPERO CRD42023424211 · 227 implants, 227 patients
Key finding: 97.8% implant survival at a weighted mean follow-up of 21.4 months across 227 CAD/CAM subperiosteal implants. Partial framework exposure occurred in 25.6% of implants the primary complication requiring attention in clinical management. Modern additively manufactured SPIs demonstrate satisfactory survival and complication rates for patients with severe alveolar atrophy.
Cairo University, Faculty of Dentistry, Egypt
Clinical and Radiographic Assessment of Milled Versus 3D-Printed Patient-Specific Subperiosteal Implants for Atrophic Mandibular Ridges: A Randomized Clinical Trial
NIH/PMC · 2025PMC11979865 · 20 patients with severely atrophic edentulous mandibular ridges · Titanium Grade 4 · Mesiodistal and buccolingual angular deviation primary outcomes
Key finding: Both CAD/CAM milled and 3D-printed patient-specific subperiosteal frameworks achieved comparable implant survival and placement accuracy, with angular deviations of less than 2° between planned and achieved framework position in both groups confirming that modern digital fabrication achieves clinically acceptable positional accuracy regardless of manufacturing method.
Policlinico Gemelli, Rome, Italy
Custom-Made 3D-Printed Subperiosteal Implants for Fixed Prosthetic Restoration of the Atrophic Posterior Mandible of Elderly Patients
Clinical Oral Implants Research · 2019PMC6950914 · 10 patients, mean age 69.6 · CBCT + DMLS titanium · 1-year follow-up · Posterior mandible partial edentulism
Key finding: Custom DMLS titanium subperiosteal implants designed from CBCT and intraoral scan data achieved predictable clinical outcomes in elderly patients with atrophic posterior mandibles over one year, providing a fixed prosthetic solution without inferior alveolar nerve repositioning or bone grafting.
Tehran University of Medical Sciences, Iran
Additively Custom-Made 3D-Printed Subperiosteal Implants for the Rehabilitation of the Severely Atrophic Maxilla
Clinical Case Reports · November 2023Arshad M, Khoramshahi N, Shirani G · DOI: 10.1002/ccr3.8135 · PMC10628114 · 3-year follow-up · Titanium alloy framework with polymer provisional
Key finding: Subperiosteal implants may represent a future first-line treatment for patients with compromised alveolar ridges. Proper CBCT-based pre-surgical imaging and digital design are required to ensure treatment success. The implant and abutments 3D-printed in titanium alloy, with a polymer provisional denture fabricated simultaneously for immediate delivery at the surgical appointment.
Frequently Asked Questions
Answers to common questions about the treatment, including suitability, procedure details, recovery, and long-term care — helping you feel informed and confident before moving forward.